Capsule and Endospore Stains
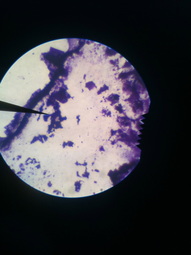 Capsule Stain Slide
Capsule Stain Slide
Today we created two different types of stains: Capsule Stain and Endospore Stain.
First we prepared a Capsule Stain. Begin with a microscope slide prepared with nigrosin as described in the preparation for a negative stain. Allow stain to dry, add crystal violet stain and gently rinse with water to remove excess stain. After blotting with bibulous paper, examine the slide under the microscope using an oil immersion lens.
From the Capsule Stain we learned that our unknown bacteria does not have a capsule.
First we prepared a Capsule Stain. Begin with a microscope slide prepared with nigrosin as described in the preparation for a negative stain. Allow stain to dry, add crystal violet stain and gently rinse with water to remove excess stain. After blotting with bibulous paper, examine the slide under the microscope using an oil immersion lens.
From the Capsule Stain we learned that our unknown bacteria does not have a capsule.
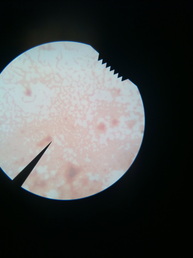 Endospore Stain Slide
Endospore Stain Slide
We next prepared an Endospore Stain. Begin preparation of stain by placing a clean microscope slide over a beaker of boiling water. Place bibulous paper on top of the microscope slide and saturate paper with malachite green stain. Let the slide with saturated paper sit for 5-6 minutes not allowing the stain to dry. After 5-6 minutes, remove slide and rinse with water for about 30 seconds to remove excess malachite green. Then proceed to cover the smear with Gram safranin stain for 60-90 seconds and rinse with water to remove excess Gram safranin. After blotting with bibulous paper, examine the microscope slide under the microscope using oil immersion lens.
From the Endospore Stain we learned that our unknown bacterium does not produce spores.
From the Endospore Stain we learned that our unknown bacterium does not produce spores.
Today we prepared four different tests to determine what our bacterium takes for nutrition. The four tests were a Starch test, Gelatin Test, Casein test, and Fat Hydrolysis Test. For the Starch, Casein and Fat Hydrolysis tests we first labeled the bottom of the plate with the appropriate test name and unknown number. Then using aseptic technique we inoculated the plate by smearing a short line on the agar surface. We then placed the plates upside down in the 37 degrees C incubator. We will return to these plates later.

Similarly, we labeled the test tube for the Gelatin test with appropriate test name and unknown number. Using aseptic technique, we inoculated the nutrient gelatin by stabbing with the inoculating needle carrying bacteria from the plate half way down the gelatin. The gelatin test tube will be incubated at 37 degrees C for forty-eight hours. We will return to this test tube later.
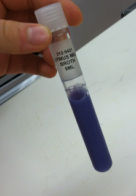
Likewise, we labeled the test tube for Litmus Milk test with appropriate unknown number. Using aseptic technique we inoculated the Litmus Milk by stirring in a loop-full of unknown bacteria. This will be incubated at 37 degrees C for a period of roughly seven days. We will return to this test tube later.

We prepared a Motility test to determine if the bacterium is motile. Obtaining an already labeled tube, we, using aseptic technique, inoculated the tube with the inoculating needle that carried bacteria from the original plate. The tube is incubated at 37 degrees C for twenty-four to forty-eight hours. We will return to this tube later.
